Background
Simulation studies have predicted a 1.8-4.-0 ºC in increase in the global average surface temperature as well as significant changes in global precipitation regimes. Climate change is expected to increase the risk of evapo-transpiration, decreased precipitation, and drier soils (Jonathan and Udall 2020). Acute drought may exacerbate stand mortality by slowly depleting the internal carbon reserves of trees referred to as carbon starvation (McDowell et al., 2008). Studies in molecular tree physiology show that drought tolerance in cellular and tissue level are led by osmotic adjustment, anti-oxidative defense and increased water use efficiency (Polle et al., 2019). Tree rings not only record historical growth rates, but also provide an archive of stable isotope ratios in tree ring cellulose over the life- time of the tree (McCarroll and Loader 2004). This study will use Carbon isotope-based analyses of tree cores from White Spruce trees from different provenances across Canada to measure water use efficiency in the various regions (Figure 3). This is based on the premise that carbon gain is closely related to moisture loss and varying populations of a species may differ in their resilience to drought for several reasons, including spatial variation in temperature and precipitation as proposed by Schwalm et al., 2017. By identifying this populations, our understanding of how tree species from within the same populations respond to drought along geographical climatic gradients will be enhanced. This knowledge will inform our selection of drought-tolerant genotypes for reforestation. The results will also provide decision support to forest managers in selecting reforestation stock that is more tolerant of drought conditions without jeopardizing growth.
What make trees resistant to drought?
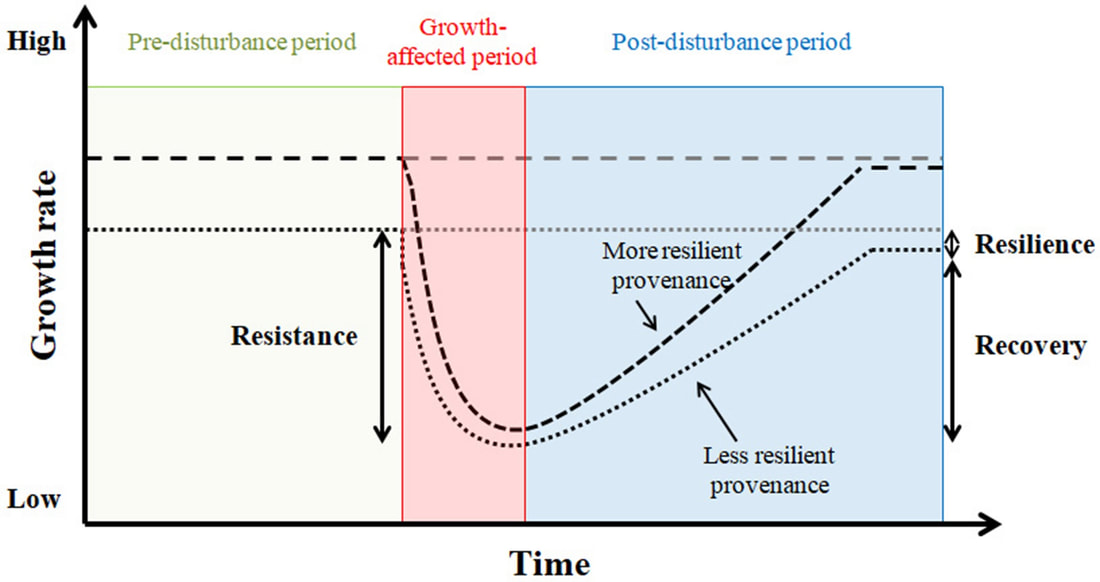
Resistance has been defined as the ability of trees to maintain their ecological functioning during a disturbance. It is expressed as the relationship between the performance during and before the disturbance (Lloret et al.,2011) as shown in Figure 1. This study considers the ratio between growth (Height and Diameter at Breast Height) and water use efficiency in the drought (dry years) and the water use efficiency in the normal years. There are many factors that make trees resistant, resilient and recover after drought occurrences. Trees resist drought conditions by closing their stomata to prevent water loss, or in other cases they have developed adaptations that allow photosynthesis with less water available in the cell, figure 2, (Yadav et al., 2016). An example of this difference is when trees regulate (close) their stomata in response to reduced water availability i.e. drought avoidance in contrast to undergoing transpiration during dry spells but selectively using tissues that are resistant to cavitation i.e. drought tolerant ( McDowell et al., 2008). Recovery is the ability of trees to recuperate in relation to the damage experienced during disturbance, and it is estimated as the ratio between performance after and during disturbance. (Lloret et al.,2011). It is usually expected that resistance to the next disturbance is determined by the rate of recovery, and this defines the long-term stability (Tilman and Downing 1994). Resilience is the potential of individuals and ecosystems to withstand a disturbance, recover from it and go back to a balanced state (Gessler et al., 2020).
Resistance has been defined as the ability of trees to maintain their ecological functioning during a disturbance. It is expressed as the relationship between the performance during and before the disturbance (Lloret et al.,2011) as shown in Figure 1. This study considers the ratio between growth (Height and Diameter at Breast Height) and water use efficiency in the drought (dry years) and the water use efficiency in the normal years. There are many factors that make trees resistant, resilient and recover after drought occurrences. Trees resist drought conditions by closing their stomata to prevent water loss, or in other cases they have developed adaptations that allow photosynthesis with less water available in the cell, figure 2, (Yadav et al., 2016). An example of this difference is when trees regulate (close) their stomata in response to reduced water availability i.e. drought avoidance in contrast to undergoing transpiration during dry spells but selectively using tissues that are resistant to cavitation i.e. drought tolerant ( McDowell et al., 2008). Recovery is the ability of trees to recuperate in relation to the damage experienced during disturbance, and it is estimated as the ratio between performance after and during disturbance. (Lloret et al.,2011). It is usually expected that resistance to the next disturbance is determined by the rate of recovery, and this defines the long-term stability (Tilman and Downing 1994). Resilience is the potential of individuals and ecosystems to withstand a disturbance, recover from it and go back to a balanced state (Gessler et al., 2020).
Water Use efficiency and drought.
Water use efficiency (WUE) estimates the relationship between plant productivity and water usage. It is usually measured by estimating the stable isotope makeup of tree rings or leaves or using eddy covariance method to measure carbon and water-fluxes (Decker et al., 2016). The carbon (δ13C) and oxygen (δ18O) stable isotope compositions in tree rings are valuable proxies for understanding the ecological and physical responses of trees to rising Carbon and changing climatic conditions over seasonal to decadal timescales (Depardieu et al., 2020). WUE of trees during photosynthesis relates to the ratio of net CO2 assimilation to transpiration rates, this infers that the greater the WUE, the more carbon is fixed per unit water lost (Farquhar et al., 1989). The carbon isotope ratio (δ13C) is expressed, in parts per thousand (‰), as
δ13C=(Rsample/Rstandard−1)1000
Where Rsample and Rstandard are the 13C/12C ratios in a sample and standard, respectively. Carbon isotopic discrimination (Δ13C) occurring in C3 plants is a result of preferential use of the lighter 12C atoms over the heavier 13C atoms during photosynthesis (McCarroll and Loader 2004). Farquhar et al.,1982b also developed a theory which predicted that this discrimination will be least in trees that have the greater WUE. However, different plant genotypes have different responses to water availability; therefore, the δ13C value is an effective index for selecting genotypes with higher WUE (Wright et al.,1994). The measurement of carbon isotope ratio (13C/12C) or carbon isotope discrimination (CID) is successfully employed as a physiological marker for high WUE in plants (Condon, 2006) due to its significantly negative correlation with WUE (Farquhar and Richards 1984). Hence higher CID is associated with lower WUE.
Disclaimer: Part of the data used in this study were fabricated for RENR 580
References
Castagneri, D., Vacchiano, G., Hacket-Pain, A. et al. Meta-analysis Reveals Different Competition Effects on Tree Growth Resistance and Resilience to Drought. Ecosystems (2021). https://doi.org/10.1007/s10021-021-00638-4
Condon, J. B. (2006). Surface Area and Porosity Determinations by Physisorption: Measurement, Classical Theories and Quantum Theory’. Elsevier.
Dekker, S. C., Groenendijk, M., Booth, B. B., Huntingford, C., & Cox, P. M. (2016). Spatial and temporal variations in plant water-use efficiency inferred from tree-ring, eddy covariance and atmospheric observations. Earth System Dynamics, 7(2), 525-533.
Depardieu, C., Girardin, M. P., Nadeau, S., Lenz, P., Bousquet, J., & Isabel, N. (2020). Adaptive genetic variation to drought in a widely distributed conifer suggests a potential for increasing forest resilience in a drying climate. New Phytologist, 227(2), 427-439.
Farquhar, G. D., O'Leary, M. H., & Berry, J. A. (1982). On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Functional Plant Biology, 9(2), 121-137.
Farquhar, G. D., & Richards, R. A. (1984). Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Functional Plant Biology, 11(6), 539-552.
Gessler, A., Bottero, A., Marshall, J., & Arend, M. (2020). The way back: recovery of trees from drought and its implication for acclimation. The New Phytologist.
Ingrisch, J., & Bahn, M. (2018). Towards a comparable quantification of resilience. Trends in Ecology & Evolution, 33(4), 251-259.
Lloret, F., Keeling, E. G., & Sala, A. (2011). Components of tree resilience: effects of successive low‐growth episodes in old ponderosa pine forests. Oikos, 120(12), 1909-1920.
McCarroll, D., & Loader, N. J. (2004). Stable isotopes in tree rings. Quaternary Science Reviews, 23(7-8), 771-801.
Polle, A., Chen, S. L., Eckert, C., & Harfouche, A. (2019). Engineering drought resistance in forest trees. Frontiers in Plant Science, 9, 1875.
Schwalm, C. R., Anderegg, W. R., Michalak, A. M., Fisher, J. B., Biondi, F., Koch, G., ... & Tian, H. (2017). Global patterns of drought recovery. Nature, 548(7666), 202-205.
Tilman, D., & Downing, J. A. (1994). Biodiversity and stability in grasslands. Nature, 367(6461), 363-365.
McCarroll, D., & Loader, N. J. (2004). Stable isotopes in tree rings. Quaternary Science Reviews, 23(7-8), 771-801.
McDowell, N., Pockman, W. T., Allen, C. D., Breshears, D. D., Cobb, N., Kolb, T., ... & Yepez, E. A. (2008). Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought?. New phytologist, 178(4), 719-739.
WrightG.C., NageswaraRaoR.C., FarquharG.D. 1994 Water use efficiency and carbon isotope discrimination in peanut under water deficit conditions. Crop Sci. 34 92– 97
Yadav, S., & Sharma, K. D. (2016). Molecular and morphophysiological analysis of drought stress in plants. Plant growth, 149-173.